What's the meaning of the Sublimation (phase transition) »
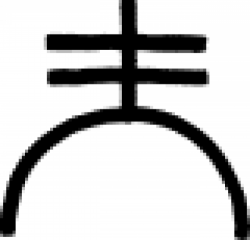
Sublimation (phase transition)
This page is about the meaning, origin and characteristic of the symbol, emblem, seal, sign, logo or flag: Sublimation (phase transition).

Sublimation is the transition of a substance directly from the solid phase to the gas phase without passing through an intermediate liquid phase.
Sublimation is an endothermic phase transition that occurs at temperatures and pressures below a substance's triple point in its phase diagram.
At normal pressures, most chemical compounds and elements possess three different states at different temperatures. In these cases, the transition from the solid to the gaseous state requires an intermediate liquid state. Note, however, that the pressure referred to here is the partial pressure of the substance, not the total (e.g., atmospheric) pressure of the entire system. So, all solids that possess an appreciable vapor pressure at a certain temperature usually can sublime in air (e.g., water ice just below 0°C). For some substances, such as carbon and arsenic, sublimation is much easier than evaporation from the melt, because the pressure of their triple point is very high, and it is difficult to obtain them as liquids.
Sublimation requires additional energy and is an endothermic change. The enthalpy of sublimation (also called heat of sublimation) can be calculated as the enthalpy of fusion plus the enthalpy of vaporization. The reverse process of sublimation is deposition. The formation of frost is an example of meteorological deposition.
Note: in alchemy, sublimation as a process automatically includes the chemical's deposition onto a portion at the top of the vessel, called a sublimate, or "flowers" because of how it often blooms into crystals.
- 2,408 Views
Graphical characteristics:
Symmetric, Open shape, Monochrome, Contains both straight and curved lines, Has no crossing lines.
Category: Alchemical Symbols.
Sublimation (phase transition) is part of the Alchemical processes group.
More symbols in Alchemical processes:
The alchemical magnum opus was sometimes expressed as a series of chemical operations. In cases where these numbered twelve, each could be assigned one of the Zodiac signs as a form of cryptography. … read more »
More symbols in Alchemical Symbols:
Alchemical symbols, originally devised as part of alchemy, were used to denote some elements and some compounds until the 18th century. Note that while notation like this was mostly standardized, sty… read more »
Citation
Use the citation below to add this symbol to your bibliography:
Style:MLAChicagoAPA
"Sublimation (phase transition)." Symbols.com. STANDS4 LLC, 2024. Web. 27 Jul 2024. <https://www.symbols.com/symbol/sublimation-%28phase-transition%29>.











Have a discussion about Sublimation (phase transition) with the community:
Report Comment
We're doing our best to make sure our content is useful, accurate and safe.
If by any chance you spot an inappropriate comment while navigating through our website please use this form to let us know, and we'll take care of it shortly.
Attachment
You need to be logged in to favorite.
Log In